Lanoxin dosages: 0.25 mg
Lanoxin packs: 60 pills, 90 pills, 120 pills, 180 pills, 270 pills, 360 pills

Lanoxin 0.25 mg buy otc
Degradation is normally as a outcome of blood pressure chart with age order 0.25 mg lanoxin with amex either hydrolysis or oxidation hypertension 140 0.25 mg lanoxin cheap free shipping, however decarboxylation, racemization and other reactions may happen. Active components known to be vulnerable to microbial assault include steroids, alkaloids, analgesics and antibiotics; several specific examples are given by Bloomfield (2007) and Baird (2011). The numbers and variety of excipients that have been reported to be degraded are no less than as nice as those of active components. Thus most categories of excipients contain materials that have been proven to be prone to microbial enzymes, acids or different metabolic merchandise. Common examples of product instability or deterioration include emulsion part separation as a outcome of surfactant degradation, lack of viscosity due to microbial effects on gums, mucilages and cellulose derivatives used as thickening brokers, and alcohol and acid accumulation following fermentation of sugars. Despite the very purpose of their use being to limit microbial development, some preservatives are susceptible to inactivation by microorganisms that, in exceptional cases, use them as a carbon and vitality source. This may lead to short-term issues for the affected person of obtaining different provides and, presumably, longer-term problems for the producer by way of buyer complaints, product recalls, adverse publicity and potential legal motion. Products and supplies weak to spoilage Spoilage, within the sense of detectable bodily or chemical change inside a pharmaceutical product, nearly at all times follows growth and copy of the contaminating organisms. The pharmacopoeial and regulatory limits for the utmost permissible numbers of microorganisms in manufactured products or raw supplies are sometimes no more than 100�1000 colony-forming items per millilitre or gram relying on the dosage kind in query. Bacteria and fungi are simply the same as all different dwelling organisms in requiring water for development (although not necessarily for mere survival). This signifies that only merchandise containing sufficient water to allow such development are weak to spoilage. Similarly, cellulosic materials, particularly paper and other packaging, might show mould development if saved in humid atmospheres. Some of the water current in an answer is bound to the solute due to hydrogen bonding or different mechanisms. Such a parameter is water activity (Aw), which is the ratio of the water vapour stress of an answer to the water vapour stress of pure water at the identical temperature. Aw is expressed on a scale from 0 to 1 � their bodily presence (cloudiness in liquid medicines, moulds on or in lotions and syrups, or as discolouration of tablets stored in a moist environment); � changes in colour (pigment production); � smell (due, for instance, to amines, acetic acid or other organic acids, or sulfides from protein breakdown); and � gas accumulation with none apparent odour (bubbles of carbon dioxide following sugar fermentation). As the concentration of solute in a solution is increased, Aw falls proportionately, and the vary of organisms able to develop in the solution progressively diminishes. Variations in water activity might arise within a single container of a manufactured medicine by, for example, water evaporating from the majority liquid during storage at high temperatures and that water vapour condensing on cool glass around the neck of a bottle as the storage temperature drops, then operating back to dilute the floor layer of the product. However, sugars and glycerol are the only common and acceptable components that could be used on this means in oral merchandise; alcohols and glycols may also be utilized in topical products. It can additionally be useful to understand how rapidly and to what focus these organisms might develop in pharmaceutical materials so as to put in place good manufacturing procedures that may decrease contamination and spoilage. Sources and kinds of contaminating organisms Microbial contamination of medicines arises from three principal sources: 1. The relative contributions of those three sources vary relying on the kind of product in question. It has been noted (see Chapter 14) that uncooked materials of various origin may differ significantly of their extent of microbial contamination. Despite the high levels of microorganisms to be present in places where many natural materials arise (gelatin, for instance, originates within the slaughterhouse, the place 853 Many widespread waterborne or soil organisms and nonskin pathogens. Pseudomonas aeruginosa, clostridia, Escherichia coli) Staphylococci and micrococci Many yeasts. This is reflected in the pharmacopoeial limit of not more than 104 colony-forming items of cardio micro organism per gram for some oral products containing supplies of natural origin compared with 102 colony-forming models per gram otherwise. Generally, the kinds of contaminating organisms are reflective of the origins of the product and this, in flip, is reflected within the objectionable organisms that should be absent. For instance, salmonellae and Escherichia coli would possibly come up in faeces, so gelatin is subject to exams for the absence of these species. The similar organisms might originate from pure fertilizers used on industrial crops, and they also should be absent too from vegetable drugs, starches, mucilages, etc. Both vegetable drugs and mined minerals may include organisms originating from the soil. Vegetable medicine could additionally be contaminated with spores of fungal plant pathogens similar to Cladosporium that rarely arise in other circumstances. Not only is it obviously current in most liquid medicines, it might be added, then removed, throughout manufacture of dry products too. It can be used in the manufacturing facility for cleaning equipment, work surfaces, mixing vessels and bottles or other product containers. As a consequence, the microbiological quality of both components and cleansing water can have a profound effect on the ultimate bioburden of the manufactured product. This is solely a consequence of the truth that mains water (potable water) is chlorinated, and the chlorine, which acts as a preservative, is removed throughout purification. Despite this purification process, purified water still contains enough dissolved vitamins to help the expansion of a quantity of species of Gram-negative bacteria to population ranges well in extra of a hundred and five per millilitre. Such levels may be attained inside days quite than weeks of room-temperature storage after chlorine removing. Product contaminants originating from the manufacturing environment all are inclined to have one attribute in frequent: they survive nicely in dry conditions. The Gram-negative micro organism that are prevalent in water are hardly ever seen in this scenario. Most of these environmental contaminants are spore-formers, both micro organism and fungi, or Gram-positive micro organism corresponding to micrococci and staphylococci. All these can persist for long intervals while hooked up to dust particles suspended within the environment or settled on floors, work surfaces or gear. The extent to which skin scales are shed is determined by many elements, such as the design and protection of protecting clothing, common well being, private hygiene and, particularly, levels of exercise. People standing or sitting usually shed far fewer particles than those who are in motion. Statistics and estimates of the extent to which humans shed skin scales differ substantially, but approximately 109 per day is a commonly quoted worth (Cosslett, 2007). Swabbing with antiseptics or washing with bactericidal soap reduces the numbers of microorganisms on the skin, however is on no account totally efficient. In the upper left section, bacterial colonies had been cultured from an unwashed finger; the higher right reveals the colonies from a finger washed with bactericidal soap and the bottom sector exhibits these from a finger swabbed for 1 minute with cotton wool soaked in antiseptic. Factors influencing the expansion of spoilage organisms In addition to water activity, which was considered earlier on this chapter, factors influencing the rate and extent of progress of a contaminant within a pharmaceutical raw material or manufactured medicine embody: � � � � � nutrient availability; temperature; pH; redox potential; and the presence and focus of antimicrobial chemicals. Some, similar to Escherichia coli, Pseudomonas aeruginosa and several Bacillus species, can synthesize all the amino acids and nutritional vitamins they want from quite lots of easy carbon and nitrogen sources. The minerals that they require are often present in enough focus as impurities in the elements of the medicinal product. Thus, in the absence of antimicrobial chemical substances, organisms of this sort could grow to concentrations of 104 per millilitre or gram, and even higher in merchandise corresponding to syrups, linctuses and lotions. Products containing glycerol, sugars, amino acids or proteins would clearly represent such ideal media for microbial progress that their preservation is typically tough to obtain regardless of the addition of preservatives. Even in the absence of those nutritionally wealthy materials, many bacteria and fungi are nonetheless in a place to utilize other parts of the formulation as meals sources.
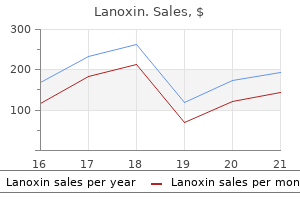
Lanoxin 0.25 mg cheap on-line
Pituitary Tumors Pituitary adenomas comprise 10 to 15% of all intracranial tumors hypertension life expectancy best 0.25 mg lanoxin. Patients have attribute endocrinological fea tures of both hypopituitarism or hypersecretion of cer tain hormones arrhythmia treatment guidelines lanoxin 0.25 mg buy with mastercard. Suprasellar extension of tumor is assessed as follows11: ��Type 0: the tumor is entirely confined inside the sella turcica. Chapter 8 Case 1: Pituitary Macroadenoma with Suprasellar Extension A 45yearold male who introduced with complaints of diminishing vision. Case 2: Pituitary Macroadenoma Fibrous Type A 35yearold girl introduced with complaints of head ache and bitemporal hemianopia. Ds, diaphragma sellae; t, tumor; mwcs-r, medial wall of right cavernous sinus; mwcs-l, medial wall of left cavernous sinus. Case three: Pituitary Macroadenoma with Suprasellar Extension A 28yearold male patient introduced with bitempo ral hemianopia. Suprasellar part of tumor dis section was carried out with with twosuction method. Case four: Revision Pituitary Macroadenoma with Suprasellar Extension A 37yearold man offered with bitemporal hemianopia and headache. Some reconstruc tive supplies which were saved in the course of the earlier sur gery were found and removed. Case 5: Revision Pituitary Macroadenoma with Suprasellar Extension A 40yearold man presented with bilateral diminishing of vision, he was operated beforehand twice for pituitary macroadenoma. As rightside nasosep tal flap was used beforehand writer decided to go for left aspect nasoseptal flap. Complete publicity of the sellar dura was done as a lot as bilateral cavernous carotid arteries. The endosteal layer of dura was incised utilizing a Cappabianca knife and sellar dural flap was elevated. Chapter eight Case 6: Pituitary Macroadenoma with Suprasellar and Right Parasellar Extension A 35yearold lady presented with historical past of complications and diminishing imaginative and prescient. T, tumor; on, optic nerve; mwcs-r, medial wall of proper cavernous sinus; mwcs-l, medial wall of left cavernous sinus. Case 7: Pituitary Macroadenoma with Suprasellar Extension A 47yearold lady introduced with bitemporal hemi anopia and headache. Chapter eight Case eight: Pituitary Macroadenoma with Suprasellar Extension A 57yearold male affected person presented with complaints of bitemporal hemianopia and headache. Chapter 8 Case 9: Pituitary Macroadenoma with Suprasellar Extension this affected person was introduced with bitemporal hemianopia. On, optic nerve; ps, planum sphenoidale; sd, sellar dura; ts, tuberculum sphenoidale. On, optic nerve; ps, planum sphenoidale; ts, tuberculum sphenoidale; sd, sellar dura; *, carotid micro Doppler. Chapter eight Pearls and Tips � In soft consistency tumors, inside debulking could be carried out utilizing two suctions inferiorly to stop early descent of the diaphragm. Fine blunt endoscopic dissection is carried out to separate fibrous bands and remove tumor in toto. Case 10: Planum Tuberculum Meningioma A 55yearold lady presented with complaints of head ache and bilateral diminishing imaginative and prescient. Superior restrict of the tumor was dissected with the assistance of neuro patties and tumor was rolled downward and once more debulked. Case eleven: Planum Tuberculum Meningioma A 35yearold woman offered with persistent head ache. Case 12: Planum Tuberculum Meningioma A 45yearold affected person introduced with bilateral diminish ing vision. Very cautious dissection of the tumor from A1 section was done with assist of pituitary elevator with out damaging it. Bilateral optic nerve, optic chiasm, pituitary gland, pituitary stalk, A1, A2, and anterior communicating arteries have been recognized. A1, a1 phase of anterior cerebral artery; a2, a2 segment of anterior cerebral artery; acom, anterior communicating artery; on, optic nerve; Pg, pituitary gland; pis, pituitary stalk. Case 13: Planum Tubercular Meningioma A 58yearold girl introduced with loss of imaginative and prescient on left aspect and diminishing imaginative and prescient on right aspect. Reconstruction was carried out in multilayer with fat, Surgicel, and nasoseptal flap. In case of adhesions to the optic nerve, cautious dissection is advised preserving the arachnoid sheet masking the blood supply of the optic pathway. In case adhesions are firm to optic nerve it is suggested to depart the adhered portion to protect functionality. The anatomical boundaries are as described for the preinfundibular kind, however due to their progress alongside the stalk, they develop along the ventral aspect of the chiasm, via the tuber cinereum and into the third ventricle. As a result, the rostral extent of the tumor is bounded by the anterior portion of the hypothalamus. They are bounded anteriorly by the stalk and posteriorly by the mammillary our bodies and basilar apex. Craniopharyngioma the resection of craniopharyngioma has excessive morbid ity as a end result of relationship with optic chiasm, pituitary stalk, hypothalamus, carotid artery, and anterior cerebral cir culation. The consistency of tumor (cystic or solid), tumor size, calcifications, and extension to the ventricles are to be taken into consideration. Many classification methods have been described to assist selec tion of surgical corridor for craniopharyngioma excision. Kassam et al and Kassam et al have categorised craniophar yngioma to information surgical approach choice (Table 8. Chapter 8 Case 14: Craniopharyngioma A 21yearold lady introduced with the history of head ache and diminishing imaginative and prescient. Debulking of the strong a part of the tumor was carried out with microdissection approach. Locr, lateral opticocarotid recess; mocr, medial opticocarotid recess; on, optic nerve; ps, planum sphenoidale; sd, sellar dura. Locr, lateral opticocarotid recess; on, optic nerve; ps, planum sphenoidale; sd, sellar dura; ts, tuberculum sellae. Cica, cavernous internal carotid artery; on: optic nerve; ps, planum sphenoidale; sd, sellar dura; *, carotid doppler. Oc, optic chiasm; on, optic nerve; t, tumor; *, anterior circulation; #, ultrasonic aspirator. Chapter 8 Case 15: Craniopharyngioma A 17yearold woman presented with headache and dimin ishing vision. Internal debulking of tumor was carried out, following which extracapsular dissection was carried out. Structures within the interpeduncular fossa had been identified similar to a basilar artery, P1 and P2 segments, and P1 perforators, and third nerve. Finally the superior half was dissected by extracapsular dissection and the third ventricle was opened up.
Diseases
- Chromosomal triplication
- Mitral valve prolapse, familial, X linked
- Quadriceps sparing myopathy
- Cyclothymia
- Ectrodactyly polydactyly
- Acquired ichthyosis
- Costochondritis (otherwise Costal chondritis)
Purchase lanoxin 0.25 mg without a prescription
Dilution and emulsification of the cream in broth containing Lubrol W blood pressure chart print lanoxin 0.25 mg order amex, polysorbate 80 or Triton X-100 might be the best process arteria radial lanoxin 0.25 mg buy generic online, though the addition of approximately 0. In practice, which means detection strategies which are described within the pharmacopoeia should be applied to a known weight of fabric (typically 1 g to 10 g), and the sample passes the check if, on the culture plates, no organisms come up that conform to the standard textbook descriptions of these to be excluded. Commercially obtainable identification kits or particular supplementary biochemical checks may be used to affirm the identity of any isolates having the everyday appearance of the goal organisms. Thus the microorganisms contained in oily merchandise have usually arisen by contamination from the environment, from tools used for manufacture and from storage vessels. To carry out a viable depend, the oil pattern should be emulsified or solubilized with out the assist of extreme warmth or some other agent that might kill the cells. An oil-in-water emulsion must be produced utilizing an acceptable surfactant; nonionic emulsifiers usually have little antimicrobial activity. Not part of European Pharmacopoeia (European Pharmacopoeia Commission, 2017) procedures. In addition, the PhEur describes a check for clostridia, but this is unlikely to be utilized to any materials aside from mined minerals. The 5 organisms common to both pharmacopoeias are the topic of those checks primarily because of their potential to trigger infections. However, they may additionally symbolize frequent contaminants of the merchandise to which the tests are utilized, or their presence could additionally be indicative of the standard of the raw material or completed manufactured product. These schemes are described in more detail elsewhere, along with images of the everyday appearance of the organisms in query (Hodges, 2000). Microbiological assays of B-group vitamins employ similar strategies to those utilized in turbidimetric assays of antibiotics (see earlier on this chapter). The extent of bacterial progress in the medium is thus immediately proportional to the quantity of reference normal or check vitamin added. It is essential to choose an assay organism that has an absolute requirement for the substance in question and is unable to acquire it by metabolism of different medium elements; species of Lactobacillus are sometimes used for this purpose. Sterile products Sterile merchandise must, by definition, be free of viable microorganisms, and it is very important understand that that is an absolute requirement. Thus, the presence of one single surviving microbial cell is sufficient to render the product nonsterile. The principal component of microbiological high quality assurance which has traditionally been applied to sterile merchandise is, of course, the test for sterility itself. In essence, this is quite easy: a pattern of the fabric to be tested is added to culture medium, which is incubated and then examined for signs of microbial progress. If progress occurs, the belief is made that the contamination arose from the sample, which consequently fails the test. However, the constraints of this simplistic method became more widely recognized within the second half of the 20th century, and there was an increasing consciousness of the reality that contaminated merchandise could cross the check and sterile ones apparently fail it (because of contamination launched in the course of the testing process itself). For these causes the sterility check alone could no longer be relied on to provide an assurance of sterility, and that assurance is now derived from a strict adherence to high quality requirements all through the manufacturing process. These encompass: � Adoption of the very best potential specifications for the microbiological high quality of the raw materials. The rationale right here is that sterilization processes usually tend to be efficient when the levels of microorganisms to be killed or removed (bioburdens) are as little as potential to begin with. Procedures used to decide bioburdens are described in Chapter 13 and earlier in this chapter. Initial validation seeks to show that adequate sterilizing situations are achieved throughout the load, and entails in depth testing with thermocouples, radiation dosimeters and biological indicators (see later) as acceptable. The pharmacopoeias and regulatory authorities require a sterility assurance stage for terminally sterilized products of 10-6 or higher. This signifies that the probability of nonsterility in an item chosen at random from a batch must be no extra than 1 in 1 million. This sterility assurance level could additionally be demonstrated within the case of some terminally sterilized merchandise just by reference to data derived from bioburdens, environmental monitoring and in-process monitoring of the sterilization process itself. Sterilization monitoring Sterilization processes may be monitored bodily, chemically or biologically (Denyer et al. Physical strategies are exemplified by thermocouples, that are routinely incorporated at totally different places within an autoclave load, whereas chemical indicators usually exhibit a color change after publicity to a heat sterilization course of. Biological indicators encompass preparations of spores of the Bacillus or Geobacillus species that displays the greatest diploma of resistance to the sterilizing agent in query. Spores of Geobacillus stearothermophilus (often still indexed in the pharmaceutical literature beneath its former name of Bacillus stearothermophilus) are used to monitor autoclaves and gaseous hydrogen peroxide or peracetic acid sterilization processes, whereas Bacillus atrophaeus is the organism normally employed for dry warmth, ethylene oxide and low-temperature steam�formaldehyde methods; Bacillus pumilus is used in radiation sterilization procedures. Spore resistance to the sterilizing agent should be carefully managed, and so rigorous standardization of manufacturing processes adopted by observance of right storage circumstances and expiry dates is important. The experimental details of those procedures are described within the PhEur (European Pharmacopoeia Commission, 2017). This section is subsequently restricted to an account of the most important options of the take a look at and a more detailed consideration of those sensible aspects that are necessary or problematical. Clearly, the results of recording an incorrect sterility outcome may be very extreme. If a fabric which was really sterile had been to fail the check, it might need to be resterilized or, more most likely, discarded. If, on the opposite hand, a contaminated batch were to pass a take a look at for sterility and be launched to be used, this would 246 clearly represent a significant health hazard. For these causes, sterility testing procedures have improved considerably lately and failures are now seen very critically by the regulatory authorities. If a product does fail, it means either that the merchandise in question is actually contaminated, by which case the manufacturing procedures are seriously insufficient, or that the merchandise is in reality sterile but the testing procedure is at fault. Sterility exams could also be conducted in clear rooms or laminar circulate cabinets which provide a grade A atmosphere as defined by the Rules and Guidance for Pharmaceutical Manufacturers and Distributors (Medicines and Healthcare merchandise Regulatory Agency, 2017). The direct inoculation technique entails the removal of samples from the product underneath take a look at and their transfer to a variety of culture media that might be anticipated to assist the expansion of contaminating organisms. After incubation, the media are examined for proof of growth, which, if current, is taken to indicate that the product may not be sterile. The measurement and variety of the samples to be taken are described within the PhEur (European Pharmacopoeia Commission, 2017). It is critical to inactivate any antimicrobial substances contained within the pattern. This different technique of conducting sterility tests is clearly solely relevant to aqueous or oily solutions that may pass by way of a membrane having a pore dimension sufficiently small to retain bacteria. The membrane, and hence the micro organism retained on it, is washed with isotonic salts resolution, which ought to remove any final traces of antimicrobial substances. This is almost invariably water as a outcome of most other frequent solvents have antimicrobial exercise. If no suitable solvent could be discovered, the broth dilution methodology is the one one obtainable.
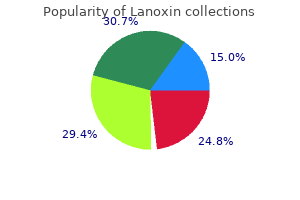
Cheap lanoxin 0.25 mg without prescription
The disintegrant most traditionally used in standard tablets is starch arrhythmia when i lay down 0.25 mg lanoxin purchase with mastercard, amongst which potato heart attack burger 0.25 mg lanoxin cheap fast delivery, maize and corn starches are commonly used. Starch particles swell in touch with water, and this swelling can subsequently disrupt the pill. However, it has also been instructed that starch particles might facilitate disintegration by particle� particle repulsion. The commonest and efficient disintegrants act through a swelling mechanism, and a collection of efficient swelling disintegrants have been developed which can swell dramatically throughout water uptake and thus rapidly and successfully break the tablet. High-swelling disintegrants are included within the formulation at comparatively low concentrations, sometimes 1% to 5% by weight. Disintegrants may be blended with different ingredients before granulation and might thus be integrated inside the granules (intragranular addition). It can also be frequent for the disintegrant to be combined with the dry granules before the complete powder combine is compacted (extragranular addition). The latter process will contribute to an effective disintegration of the tablet into smaller fragments. A third group of disintegrants function by producing fuel, usually carbon dioxide, when in touch with water. Such disintegrants are used in effervescent tablets and usually not in tablets that should be swallowed as a strong. The liberation of carbon dioxide is obtained by the decomposition of bicarbonate or carbonate salts in contact with acidic water. The acidic pH is accomplished by the incorporation of a weak acid in the formulation, similar to citric acid or tartaric acid. Dissolution enhancer For medicine of low aqueous solubility, the dissolution fee of the drug could be the rate-limiting step in the general drug release and absorption processes. Agents other than matrix formers may due to this fact generally be discovered within the composition of a tablet with the role to speed up the drug dissolution course of by briefly growing the solubility of the drug during drug dissolution. An important example of a dissolution enhancer is the incorporation into the formulation of a substance that types a salt with the drug throughout dissolution. As mentioned already, a surfactant may facilitate wetting of hydrophobic drug particles and enhance the surface area available for drug dissolution. A surfactant may also increase the rate of dissolution of poorly soluble drugs via a solubilization process. This may be conceptually described by method of the formation of micelles in vivo adopted by the dissolution of the drug into the micelles, increasing the apparent drug solubility. Absorption enhancer For medication with poor absorption properties, the absorption can be affected (see Chapter 20) by utilizing substances in the formulation that affect the permeability of the intestinal cell membrane, and thus increase the speed at which the drug passes though the intestinal membrane. An additive that modulates the permeability of the gut is commonly referred to as an absorption enhancer. Glidants are used in formulations for direct compaction but are often also added to granules before tableting to be certain that sufficient flowability of the pill mass is achieved for prime production speeds. Traditionally, talc has been used as a glidant in pill formulations, in concentrations of approximately 1% to 2% by weight. Today, the most generally used glidant is probably colloidal silica, added in very low proportions (approximately zero. Because the silica particles are very small, they adhere to the particle surfaces of the other elements. Magnesium stearate, usually used as a lubricant, also can promote powder circulate at low concentrations (< 1% by weight). Binder A binder (also generally known as an adhesive) is added to a drug�filler mixture to be certain that granules and tablets may be shaped with the required mechanical energy. Binders can be added to a powder in numerous methods: � As a dry powder which is mixed with the opposite ingredients before wet agglomeration. During the agglomeration process, the binder might thus dissolve partly or completely in the agglomeration liquid. Both solution binders and dry binders are included within the formulation at relatively low concentrations, sometimes 2% to 10% by weight. Polymers such as polyvinylpyrrolidone and cellulose derivatives (in explicit hydroxypropyl methylcellulose), with improved adhesive properties, are more commonly used binders today. Microcrystalline cellulose and cross-linked polyvinylpyrrolidone are necessary examples of dry binders. Solution binders are typically considered the best, and their use is therefore the most common way of incorporating a binder into granules; the granules thus formed are sometimes referred to as binder� substrate granules. High friction throughout tableting may cause a sequence of problems, including insufficient tablet quality (capping and even fragmentation of tablets during ejection and vertical scratches on pill edges), and will even stop production. In fluid lubrication a layer of fluid is positioned between and separates the transferring surfaces of the solids from each other and thus reduces the friction. However, liquid paraffin has been used, as an example in formulations for effervescent tablets. Boundary lubrication is considered a surface phenomenon, as right here the sliding surfaces are separated by solely a really skinny movie of lubricant. In boundary lubrication the friction coefficient and put on of the solids are greater than with fluid lubrication. The lubricants used in tablet formulations appearing by boundary lubrication are fantastic particulate solids. A number of mechanisms have been discussed for these boundary lubricants, including that lubricants are substances which have a low resistance to shearing. Magnesium stearate has turn into the most widely used lubricant owing to its superior lubrication properties. Besides lowering friction, lubricants might trigger undesirable adjustments in the properties of the tablet. Because many lubricants are hydrophobic, tablet disintegration and dissolution are often retarded by the addition of a lubricant. These adverse effects are strongly associated to the amount of lubricant current, and a minimum amount is normally utilized in a formulation, i. In addition, the way by which the lubricant is mixed with the other ingredients also wants to be thought-about. It can, for instance, be essential if the excipients are added sequentially to a granulation quite than concurrently. The whole mixing time and the blending depth are also essential on this context. The generally observed retardation of disintegration and dissolution of tablets is said to the hydrophobic character of the most commonly used lubricants. In order to avoid these adverse effects, more hydrophilic substances have been instructed as alternate options to the hydrophobic lubricants.

Lanoxin 0.25 mg generic line
Commonly used buffers in ophthalmic solutions embody borate and phosphate buffers heart attack jogging 0.25 mg lanoxin cheap amex. To prepare solutions of decrease pH blood pressure for teens cheap lanoxin 0.25 mg without prescription, acetic acid/sodium acetate and citric acid/sodium citrate buffers are used. Surface rigidity the surface tension of tear fluid at physiological temperature in a healthy eye is 43. Administration of options which have a surface tension much lower than that of the lacrimal fluid destabilizes the tear movie and disperses the lipid layer into droplets which might be solubilized by the drug or surfactants within the formulation. Surfactants are sometimes included in ophthalmic preparations to solubilize or disperse medication. Nonionic surfactants are the least irritant and due to this fact essentially the most generally used; examples embody polysorbate 20, polyoxyl forty stearate and polyoxypropylene� polyoxyethylenediol. Despite being the least irritant, nonionic surfactants have been shown to take away the mucous layer and disrupt the tight junction complexes of the cornea, thereby growing drug permeation. Surfactants can also work together with polymeric substances within the preparation and scale back the efficacy of preservatives. The focus of surfactant is necessary not only when it comes to drug solubility, security and patient tolerance, but also as a result of high concentrations can lead to foaming on product manufacture or shaking. Hydrogen ion focus (pH) the pH of tears is near impartial and is controlled by numerous substances dissolved within the aqueous layer of tears: carbon dioxide, bicarbonate, proteins, enzymes and fatty acids. The eye can usually tolerate topical ophthalmic preparations at a pH throughout the range of three. It undergoes pH-dependent hydrolytic degradation, and one of many methods to obtain stability of pilocarpine aqueous answer is to maintain the pH at 3. This is often seen with linear, multiple-charged polymers corresponding to sodium hyaluronate and carbomers. Acceptable viscosity of ophthalmic preparations is up to 15 mPa s; past that increased lacrimation and drainage occur to restore the tear film to its physiological viscosity. Furthermore, very viscous solutions can cause blurring of vision, potential blocking of the puncta and canaliculi, and pain on blinking. Topical, liquid ophthalmic preparations Solutions Ophthalmic solutions are the commonest topical ophthalmic preparation. They are typically the best to manufacture (have the lowest manufacturing cost) and are relatively simple for a patient or well being care provider to administer. Moreover, options are homogeneous and subsequently show a greater dose uniformity. Moreover, the rate of drainage is proportional to the scale of the drop administered. The quantity of eye drops administered from industrial eye dropper bottles has been reported to be within the vary of 34 �L to sixty three �L; that is depending on the physical shape and orifice of the dropper opening, the physicochemical properties of the liquid and the manner during which the dropper is used. This method has been used to administer drugs that are sparingly soluble in water. Particles tend to be retained within the conjunctival sac (pouch the place the conjunctiva masking the within of the decrease eyelid and the sclera meet) and slowly go into resolution, thus rising the contact time. Particle measurement and shape have to be rigorously selected as some particles can cause irritation of the sensory nerves within the epithelium. The European Pharmacopoeia and United States Pharmacopeia set limits for the maximum particle dimension permitted in ocular suspensions as a end result of giant particles give rise to irritation and increased tearing. The particles of a suspension have to be readily dispersible on shaking by the affected person to ensure uniform dose administration. Homogeneity and dose uniformity have to be confirmed in multidose containers from first to final use. A problem that may arise with suspensions is conversion of the crystal structure of the drug. If the drug particle size is polydisperse, Ostwald ripening might happen on changes in storage temperature or prolonged storage. Cake formation can be an issue, which is most likely not resolved by forming a flocculated suspension since giant floccules can irritate the attention. Using a polymer solution as a viscosity-enhancing agent can prevent caking and allow particle resuspension by shaking. Nepafenac is a nonsteroidal anti-inflammatory prodrug indicated for the remedy of pain and inflammation associated with cataract surgery. It is practically insoluble in water and is subsequently formulated as suspension formulations: Nevanac 0. The newer Ilevro formulation has a higher focus of the active substance, 2. These formulation modifications have increased ocular bioavailability and allowed once every day administration, thus improving affected person convenience/ adherence. It has been successfully solubilized in an oil-in-water submicron emulsion formulated at a pH of 6. The oil phase in Restasis is castor oil and the emulsion is stabilized with the nonionic surfactant polysorbate eighty and glycerine, which behaves here as a cosurfactant. Topical, semisolid ophthalmic preparations Ointments Ophthalmic ointments represent approximately 10% of ophthalmic products and are often used for the treatment of inflammation, infections and ocular floor illness. They offer the benefit of decreasing drug drainage by tear circulate, thereby growing corneal residence time. Ointments can also be entrapped in the conjunctival sac, whereby they serve as a reservoir for the drug. Ointments also have the benefit of permitting the incorporation of drugs with poor aqueous solubility. Hydrophobic ointments generally enhance the soundness of hydrolysable compounds, particularly peptides. Soft paraffin and liquid paraffin are commonly used as bases for ophthalmic ointments. Antibiotics, antifungals and steroids are the classes of medicine most out there as ointments. Drug bioavailability normally peaks later with ointment automobiles than with solutions or suspensions. Ointments are, nevertheless, more difficult to administer than options and will give rise to a extra variable dose. In addition, blurring of imaginative and prescient arises, which tends to cut back affected person adherence, making ointments more useful for night-time administration. Drug molecules could additionally be entrapped 698 within the ointment base because of beneficial partitioning in course of the base, subsequently inhibiting drug release. Gels Gels, which are semisolid techniques comprising water-soluble bases, are additionally out there and are extra favourable than ointments for water-soluble drugs. Ganciclovir is an antiviral indicated for herpetic keratitis (eye ulcers brought on by herpes virus which mainly have an effect on the cornea). These undergo a part transition from liquid to solid in the conjunctival sac to form a viscoelastic gel.
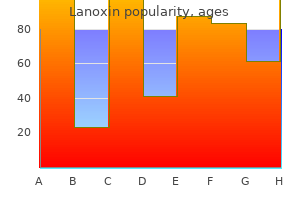
Buy discount lanoxin 0.25 mg on line
Hence medication which would possibly be substrates for P-gp will be much less well absorbed throughout the nasal epithelium than their physicochemical properties (molecular dimension blood pressure normal low pulse buy 0.25 mg lanoxin otc, lipophilicity blood pressure 14080 buy 0.25 mg lanoxin mastercard, diploma of ionization) might predict. Active transport/efflux operates in opposition to a focus gradient, is saturable and may be competitively inhibited by different substrates for the binding website. Thus coadministration of an inhibitor of P-gp, such as rifampicin or verapamil, can improve drug absorption. P-gp can additionally be discovered in the olfactory epithelium, at a higher concentration 678 Solubility the strategies to improve the solubility of a drug can contain (1) number of a unique salt type, (2) modification of its molecular type (including the usage of a prodrug) and (3) using appropriate excipients, similar to cosolvents, when the drug is formulated (considered later). Prodrugs are often developed to increase the lipophilicity of a drug molecule and hence its absorption throughout a organic membrane. However, within the case of nasal supply, the precept has been explored to improve the aqueous solubility of the parent drug to enable a clinically relevant dose of the drug to be dissolved in lower than a hundred and fifty �L of resolution, and has been profitable for several medicine. The prodrug is quickly converted to the energetic mother or father drug once it enters the bloodstream. The appropriate selection of the salt form of an ionizable drug can be utilized to improve its aqueous solubility. However, a change in salt type can outcome in irritancy to the nasal mucosa and this has to be thought of when an acceptable counterion is being chosen. The ionized type of the drug also reveals some permeation capability, the diploma of which may be dependent on the nature of the counterion. Lipophilicity/hydrophilicity and molecular measurement Once in solution, lipophilic medication such as propranolol, progesterone and fentanyl are quickly absorbed from the nasal cavity by the transcellular route and have a nasal bioavailability just like that obtained after intravenous administration (almost 100%). The absorption of hydrophilic (polar) medicine occurs through the paracellular route (between the epithelial cells through the tight junctions), and the speed and extent of absorption is inversely proportional to the molecular weight of the drug. The paracellular route supplies a much smaller space for absorption than the transcellular route. Thus the absorption of hydrophilic compounds is far slower than that of lipophilic medication. For both lipophilic and hydrophilic molecules, absorption is relatively environment friendly for medicine with a molecular mass beneath 1 kDa however then declines as molecular mass increases. With regard to dose reproducibility from the nasal cavity, dosing is comparatively constant for low molecular weight medicine in comparison with the oral or parenteral routes, whereas for compounds with a high molecular weight, corresponding to peptides and proteins, relatively excessive variability is exhibited in contrast with injections. Formulation components affecting intranasal systemic supply the identical basic formulation issues apply to medication formulated for systemic motion as for native action, as indicated by the examples shown in Table 38. However, extra methods can be utilized to improve absorption throughout the nasal epithelium. In essence, the bioavailability of nasally administered medication can be limited by: � low aqueous solubility; � speedy and in depth enzymatic degradation of the drug within the nasal cavity; � quick contact time between the drug and the absorptive epithelium of the turbinates because of mucociliary clearance; and � poor permeation of the drug throughout the respiratory epithelium. The approaches which were used to overcome these limitations are summarized in Table 38. Increasing aqueous solubility As discussed already, for a drug to be absorbed, it should normally be in answer. Drug solubility may be elevated by use of a blended solvent system or a cosolvent in the formulation. Cyclodextrins (see Chapter 24) are cyclic compounds composed of -D-glucopyranose items. They tend to be water soluble due to their hydrophilic/ polar outer surface, but have a hydrophobic/less polar 679 Degree of ionization For medication which may be weak acids or bases, the pH of the nasal cavity will affect the degree of ionization of the drug. In addition, the pH of the formulation itself can alter the native pH, particularly if buffered vehicles are used. They are capable of increase the aqueous solubility of lipophilic compounds by forming dynamic inclusion complexes where the lipophilic a part of the drug molecule is included into the lipophilic central cavity of the cyclodextrin ring. An intranasal formulation containing 17-estradiol solubilized in dimethyl-cyclodextrin (seven glucopyranose units) was obtainable for the treatment of menopausal symptoms, till it was withdrawn in 2006. The formulation was well-tolerated and as effective as transdermal and oral formulations of estradiol. The dimethyl-cyclodextrin was reported to improve absorption of the drug by both enhancing its solubility and rising the permeability of the nasal epithelium. Proteolytic enzyme inhibitors could prevent the hydrolysis of peptide and protein medicine within the nasal cavity, bettering their stability at the absorption web site. As examples, the aminopeptidase and trypsin inhibitor camostat mesilate increased the nasal absorption of the peptide vasopressin and its analogue desmopressin, and the absorption of calcitonin can be enhanced by the use of trypsin inhibitors. The pH of a formulation is mostly dictated by the soundness of the drug however, within these constraints, a pH favouring extra unionized molecules would be expected to enhance absorption. It is necessary to acknowledge that the formulation should be nonirritant to the nasal mucosa, and formulation at a pH near that of the nasal cavity (5. Polymeric materials can adhere to each the nasal epithelial floor (bioadhesion) and nasal mucus (mucoadhesion). Mucoadhesive formulations can be administered to the nasal cavity within the form of solid powders or particulates, gels or liquids. For good mucoadhesion, the formulation ought to spread nicely on the nasal mucosa (solid formulations ought to circulate well and be readily wettable), after which the hydration of the polymer and the intimate contact it has with the nasal mucosa is very important. Mucoadhesives can enhance absorption by three mechanisms: formulation by mucociliary clearance, thus increasing its retention time in the nasal cavity. As the polymer becomes hydrated, it causes dehydration of the epithelial cells, which might briefly open the tight junctions, so rising permeability of the epithelium with regard to medicine utilizing the paracellular route. With time, the continuous production of mucus will trigger additional hydration of the polymer (beyond the optimum required for mucoadhesion), the energy of mucoadhesion will diminish and normal mucociliary clearance will resume, so clearing the polymer from the nasal cavity. Examples of polymers and medicines which were used in studies of nasal mucoadhesion are given in Table 38. When the polymers are formulated in solution, the viscosity of the preparation shall be larger than that of a simple solution. Whilst an � Optimum hydration will promote the extension of polymer chains which will interact with the nasal tissue and resist the removing of the Table 38. This could be as a result of the decreased fee of diffusion of the drug molecules via an answer of higher viscosity. In studies, this approach has been used efficiently to improve the nasal absorption of metoclopramide, sumatripan and insulin. Fentanyl is a low molecular weight, lipophilic molecule that readily crosses the nasal epithelium and is beneficial for the therapy of breakthrough ache, with a extra rapid onset of action and higher bioavailability from the nasal cavity than from the oral transmucosal route (buccal or sublingual). Powders have certain advantages over liquid formulations, and these include: attainable irritation of the nasal mucosa and a attainable gritty texture. The aerodynamic dimension of the particles (see Chapter 37) will affect the deposition web site within the nasal cavity, and manufacturing particles of the right aerodynamic particle size to deposit within the respiratory region of the nasal cavity, where most absorption happens, could be costly. Polymers may additionally be formulated as microparticles/ microspheres and nanoparticles (see Chapter 44).
Cuscutae (Dodder). Lanoxin.
- Bladder, liver, and spleen problems.
- Are there safety concerns?
- Dosing considerations for Dodder.
- How does Dodder work?
- What is Dodder?
Source: http://www.rxlist.com/script/main/art.asp?articlekey=96067
Purchase lanoxin 0.25 mg overnight delivery
Drug dissociation and lipid solubility the dissociation constant and lipid solubility of a drug and the pH at the absorption site typically influence the absorption traits of a drug all through the gastrointestinal tract blood pressure chart software free 0.25 mg lanoxin discount visa. As most medicine are weak electrolytes blood pressure medication bananas lanoxin 0.25 mg buy without prescription, the un-ionized form of weakly acidic or primary drugs. Consequently, based on the pH-partition speculation, the absorption of a weak electrolyte will be determined mainly by the extent to which the drug exists in its un-ionized kind on the website of absorption. The extent to which a weakly acidic or primary drug ionizes in solution in the gastrointestinal fluid could also be calculated using the appropriate form of the Henderson�Hasselbalch equation (discussed further in Chapter 3). However, in apply, very little absorption happens in the abdomen and lots of different components must be considered. Despite their excessive diploma of ionization, weak acids are still quite nicely absorbed from the small intestine. In truth, the rate of intestinal absorption of a weak acid is commonly larger than its price of absorption within the abdomen, despite the fact that the drug is un-ionized within the stomach. In addition, a longer small intestinal residence time and a microclimate pH (which exists at the surface of the intestinal mucosa and is lower than that of the luminal pH of the small intestine) are thought to assist the absorption of weak acids from the small intestine. During absorption, drug molecules should diffuse throughout this layer and then on via the lipid layer. Diffusion throughout this layer is liable to be a major factor of the total absorption course of for these drugs that cross the lipid layer very quickly. The movement of water molecules into and out of the gastrointestinal tract will have an result on the speed of passage of small water-soluble molecules across the gastrointestinal barrier. Water movement occurs due to variations in osmotic pressure between blood and the luminal contents and due to differences in hydrostatic pressure between the lumen and the perivascular tissue. The absorption of watersoluble medicine might be increased if water flows from the lumen to the blood, offered that the drug and water are utilizing the identical route of absorption. This may have the greatest impact within the jejunum, where water movement is at its greatest. Smaller molecules which are poorly lipid soluble and hydrophilic in nature, such because the -blocker atenolol, could be absorbed by way of the paracellular route. Drugs which are very lipid soluble (log P > 3) are most likely to be nicely absorbed however are additionally more prone to be vulnerable to metabolism and biliary clearance. A prodrug is a chemical modification, regularly an ester of an current drug, which converts again to the parent compound as a result of metabolism by the body. A Lipid solubility A number of drugs are poorly absorbed from the gastrointestinal tract regardless of their un-ionized types predominating. For example, the barbiturates barbitone and thiopentone have comparable dissociation constants � pKa of 7. The cause for this difference is that the absorption of medication is also affected by the lipid solubility of the drug. Thiopentone, being more lipid soluble than barbitone, has a higher affinity for the gastrointestinal membrane and is thus far better absorbed. An indication of the lipid solubility of a drug, and therefore whether that drug is liable to be transported across membranes, is given by its ability to partition between a lipid-like solvent and water or an aqueous buffer. Active drug Ampicillin Ampicillin Carbenicillin Cefuroxime Enalaprilat Terbutaline Aciclovir Amprenavir Ester Pivaloyloxymethyl Carbonate Indanyl Acetylethyl 1-Carboxylic acid Dibutyl L-Valyl Dosage type components influencing bioavailability Introduction the fee and/or extent of absorption of a drug from the gastrointestinal tract has been shown to be influenced by many physiological elements and by many physicochemical properties associated with the drug itself. The bioavailability of a drug may also be influenced by elements related to the formulation and production of the dosage type. Increasingly, many dosage forms are being designed to have an result on the discharge and absorption of medicine; for example, controlledrelease methods (see Chapter 31) and delivery methods for poorly soluble medication. This part summarizes how the kind of dosage kind and the excipients utilized in conventional oral dosage varieties can have an effect on the speed and extent of drug absorption. Examples of prodrugs which have been successfully used to increase the lipid solubility and therefore absorption of their parent drugs are proven in Table 20. Molecular dimension and hydrogen bonding Two other drug properties which would possibly be essential in permeability are the variety of hydrogen bonds throughout the molecule and the molecular dimension. For paracellular absorption, the molecular mass should ideally be lower than 200 Da; nevertheless, there are examples where larger molecules (with molecular plenty up to 400 Da) have been absorbed through this route. In general, for transcellular passive diffusion, a molecular mass of lower than 500 Da is preferable. There are few examples of medication with molecular lots larger than seven hundred Da being well absorbed. In general, not extra than 5 hydrogen-bond donors and no extra than 10 hydrogen-bond acceptors (the sum of the number of nitrogen and oxygen atoms in the molecule is often taken as a tough measure of the variety of hydrogenbond acceptors) must be present if the molecule is to be properly absorbed. The massive number of hydrogen bonds inside peptides is probably certainly one of the reasons why peptide medication are poorly absorbed. The type of oral dosage kind will affect the variety of potential intervening steps between administration and the appearance of dissolved drug in the gastrointestinal fluids, i. In basic, medicine have to be in answer within the gastrointestinal fluids before absorption can happen. Thus the larger the number of intervening steps, the greater will be the variety of potential obstacles to absorption and the higher would be the probability of that sort of dosage type reducing the bioavailability exhibited by the drug. Hence the bioavailability of a given drug tends to lower in the following order of the kinds of dosage kind: aqueous solutions > aqueous suspensions > stable dosage forms. Aqueous options For medicine which are water soluble and chemically stable in aqueous answer, formulation as an answer usually eliminates the in vivo dissolution step and presents the drug in the most available form for absorption. However, dilution of an aqueous solution of a poorly water-soluble drug whose aqueous solubility had been increased by formulation techniques similar to cosolvency, complex formation or solubilization can lead to precipitation of the drug within the gastric fluids. Similarly, exposure of an aqueous resolution of a salt of a weak acidic compound to gastric pH can also result in precipitation of the free acid form of the drug. In most circumstances the extraordinarily fantastic nature of the resulting precipitate permits a extra fast price of dissolution than if the drug had been administered in other kinds of oral dosage varieties, corresponding to an aqueous suspension, onerous gelatin capsule or tablet. However, for some medicine this precipitation can have a significant effect on bioavailability. For example, the identical dose of an experimental drug was given to canine in three totally different solution formulations: a polyethylene glycol answer and two completely different concentrations of hydroxypropyl-cyclodextrin. Bioavailabilities of 19%, 57% and 89% 330 had been obtained for polyethylene glycol, the lower concentration of hydroxypropyl-cyclodextrin and the upper focus of hydroxypropyl-cyclodextrin respectively. The distinction in bioavailability of the three options was attributed to the distinction in the rates of precipitation of the candidate drug from the three solutions on dilution. The experimental drug was observed to precipitate most rapidly from the polyethylene glycol solution, and most slowly from the most concentrated hydroxypropyl-cyclodextrin solution. Factors related to the formulation of aqueous options that can affect drug bioavailability embrace: � the chemical stability exhibited by the drug in aqueous solution and the gastrointestinal fluids. The formation of such a posh can enhance the aqueous solubility of the drug, which may increase bioavailability or enhance the viscosity of the dosage type, which might have a detrimental impact on bioavailability. Information concerning the potential affect of each of those elements was given earlier in this chapter. Aqueous suspensions An aqueous suspension is a useful dosage kind for administration of an insoluble or poorly water-soluble drug. Usually the absorption of a drug from this type of dosage type is dissolution-rate restricted. The oral administration of an aqueous suspension ends in a large whole floor space of dispersed drug being instantly presented to the gastrointestinal fluids.
Purchase lanoxin 0.25 mg visa
The European Pharmacopoeia classifies glass containers relying on the composition of the glass arterial line lanoxin 0.25 mg buy generic, any floor remedy and its hydrolytic resistance can high blood pressure medication cause joint pain 0.25 mg lanoxin buy with visa. Microbial spoilage and the preservation of pharmaceutical products are thought of in Chapter forty eight. Stability testing of pharmaceutical merchandise the purpose of stability testing is to provide proof on how the standard of a drug substance or drug product varies with time under the affect of a selection of environmental elements, such as temperature, humidity and light-weight, and to set up a retest period for the drug substance or a shelf life for the drug product and really helpful storage situations. The stability of a pharmaceutical product is complex, usually being dependent on a quantity of physical, chemical and microbiological components which will or might not work together with one another, such that an appropriate stability profile can by no means be fully predicted � in brief, this complexity implies that stability testing of a pharmaceutical product is a necessary part of any development programme. Microbiological stability Proliferation of bacteria, yeasts and moulds in a drug product can adversely affect the product itself, as properly as pose a severe danger to affected person safety. Pharmacopoeias lay down requirements for the microbial quality of each sterile and non-sterile products. Other injectable merchandise, such as insulin vials, and eye drops could include a preservative system permitting 870 Types of stability studies Preformulation research In the preformulation stage, the physicochemical properties of the lively substance are fully characterised (see Chapter 23). Stress testing or compelled stability research expose the active substance, both stable and solubilized forms, to excessive circumstances over a short interval. Conditions should be selected to obtain a degradation level of roughly 10%, although this level may range from one firm to another, and from one active substance to one other. The purpose of these research is to degrade the lively substance to elucidate the probably degradation mechanisms and pathways, which is useful for the development of the container closure system and the formulation of the drug product. Of course, where an existing lively substance is used, this information may be derived from published literature sources. Stress testing or pressured stability studies are additionally used to validate and demonstrate that the analytical methods are stability indicating. Formulation and container improvement stability research A drug product may bear several phases of formulation growth. The first formulation stage could additionally be for preclinical studies or part I (first in humans) medical trials, where the product may be a easy parenteral injection or an early prototype formulation. Stability testing of the completely different formulations is carried out at applicable stages to assist the event programme, in addition to to present stability data to assist the clinical trial (regulatory authorities require demonstration that scientific trial supplies are appropriately secure and are safe to be administered to volunteers). The forms of stability study can vary from forced degradation or stress testing of the drug product (principally to validate the analytical methods as stability indicating, however also can provide useful information on potential interactions with excipients within a formulated product), accelerated testing (which may give a sign of the probably long-term stability of different development formulations) and long-term or real-time testing (on prototype laboratory-scale or small pilot scale improvement batches) to formal stability studies (Table 49. Temperature biking research (from lower than 0 �C to forty �C) should be performed for inhalation, nasal and transdermal merchandise, in addition to suspensions, lotions and emulsions (even solutions, in some cases). Temperature fluctuations can encourage precipitation and/or particle progress, which might have a major impact on the standard and efficacy of these merchandise. The performance of inhalation and nasal products at low temperature additionally needs to be characterised. Binary mixes of the energetic substance and excipient can then be used to affirm excipient compatibility. Binary mixes are normally a 1: 1 ratio of the active substance and excipient in either a powder blend/compact or an aqueous slurry mixture (to investigate the impact of moisture), though it could be more scientific for the ratio to mimic the degrees more likely to be used in the formulation. Tertiary mixes, where an extra excipient is added to the binary mix, may be used. Alternatively, thermal methods, such as differential scanning calorimetry, can be used to detect interactions between the lively substance and excipients. The manufacturer must choose the most appropriate sort of study to achieve the utmost amount of helpful data on the steadiness of the drug product, while making essentially the most environment friendly use of the formulation, manufacturing, analytical, stability cabinet storage, financial and time resources at its disposal. Stability testing types a big a half of a improvement programme each financially and in time, so it could be very important get it proper first time. Pharmaceutical merchandise are likely to bear many modifications during their lifecycle. It is sort of frequent for producers to amend the drug product formulation or change the manufacturing course of and tools, change suppliers of lively substances/ excipients, change the producer of the drug product or change the container closure system. All these adjustments might need to be submitted to the regulatory authorities and supported by stability research if a variation is sought to the advertising authorization. This requires manufacturers to provoke an ongoing or rolling stability programme, by which no less than one batch per yr of product manufactured, of each strength and each main packaging sort is included, unless otherwise justified. The stability examine design can be totally different from that used for licensing purposes, however should generate adequate stability knowledge to allow development analysis in order that adjustments in the stability profile can be detected. The stability of the product during transportation from the manufacturer(s) to the wholesaler(s) and eventually the pharmacy also needs be verified, because the recommended storage advice applies to each static and cellular (during transportation) storage (Medicines and Healthcare products Regulatory Agency, 2017). However, Schumacher (1972) and Grimm (1986, 1998) proposed lowering the variety of long-term take a look at conditions on the premise of the environmental situations in just four climatic zones. The current proposed meteorological criteria for the totally different climatic zones are described in Table forty nine. Climatic conditions can differ between the different areas, and can change all year long. It is higher than the arithmetic mean temperature, and takes into account the Arrhenius equation. These storage conditions describe the final case for testing pharmaceutical merchandise. If the product is intended to be stored in a refrigerator, the long-term storage condition is 5 �C � 3 �C and accelerated storage is 25 �C/65% relative humidity, 30 �C/65% relative humidity or 30 �C/75% relative humidity. Products meant for storage in a freezer have a long-term situation of -20 �C; products that need to be saved under -20 �C are handled on a case-by-case foundation. Alternatively, the next storage humidity can be used, and water loss at the reference relative humidity could be derived via calculation. At forty �C, the calculated fee of water loss during storage at no more than 25% relative humidity is the same as the measured water loss fee at 75% relative humidity multiplied by three. Testing at accelerated and intermediate circumstances Accelerated circumstances are designed to be a moderately extra annoying temperature and relative humidity setting than the long-term storage conditions, with intermediate circumstances somewhere within the middle. Accelerated situations should be differentiated from stress testing, the place more excessive circumstances may be used. Pharmaceutical merchandise are typically steady (in the order of years) at long-term storage circumstances, and thus stability testing over this era presents a sensible downside for the manufacturer. Testing at accelerated or intermediate situations can significantly reduce the time taken to generate stability data, giving an early indicator of the stability of the pharmaceutical product. Accelerated or intermediate stability knowledge can be used within the extrapolation of the out there long-term stability data to set longer retest intervals or shelf lives than the period coated by the long-term data. It is because of this primarily that it is suggested to initiate stability testing at accelerated and/or intermediate circumstances, along with long-term conditions, in any formal stability research design. To cut back this uncertainty, the concepts of a moisturecorrected Arrhenius equation (Eqn. This overcomes some of the limitations of the empirically (experimentally) derived Arrhenius equation being unable to precisely replicate the complicated individual molecular reactions concerned in degradation (as described earlier). For example, in a stable dosage kind, active substance molecules can exist in an amorphous state or in crystalline domains, and could additionally be adjoining to different excipients. Consequentially, the molecules might degrade at completely different rates as a operate of the amount of degradation.
0.25 mg lanoxin generic visa
The enhance within the variety of individual expenses inside the diffuse layer will end in easier neutralization of the remaining cost from the particle and therefore will lead to hypertension juice recipe cheap lanoxin 0.25 mg with amex a thinning of the diffuse layer blood pressure kits for sale generic 0.25 mg lanoxin visa. It ought to be evident from the earlier discussion that an elevated focus of the additional costs would be anticipated to result in a larger discount of 1/. In reality, the connection is a sq. root one, in that 1/ is inversely proportional to the sq. root of the ionic strength of the medium. This is the same as saying that (the Debye�H�ckel size parameter; see Chapter 5) is instantly proportional to the square root of the ionic power of the medium. Care have to be taken, therefore, to think about the chemistry of dissolved ionic supplies: the ionic power of a calcium chloride (CaCl2) solution is higher than that of a sodium chloride (NaCl) solution of the same molar focus. The effects of these two solutions on the electrical double layer will consequently be totally different. In this case the cost on the particle floor will decrease, which may have the automatic effect of reducing the Stern potential and a secondary effect of reducing the zeta potential, because the charge discount throughout the diffuse layer will begin at a unique worth. At concentrations above the cmc, surfactant micelles might be fashioned, with a central hydrophobic core into which the hydrophobic drug could dissolve (see Chapters 5 and 24). The addition of the surfactant to the floor of the particle will change the particulate cost, certainly in its magnitude however probably additionally in its signal. Such cost modification will have an effect on the mounted layer instantly, somewhat than the diffuse layer, and a variation within the surface charge will naturally result in an alteration of the Stern potential. As described earlier, this can then have a secondary effect on the zeta potential, because the cost decay across the diffuse layer will begin from a unique value. Na+ ions from sodium lauryl sulfate), which will then have their own direct effects on the diffuse layer. The total impact of addition of surfactants might need to be thought of on a case-by-case foundation, on the basis of their chemistry. These interactions can, to some extent, be regarded as the interactions of the diffuse layers around particular person particles, and therefore the electrical double layer supplies the premise for understanding interparticulate interactions. Essentially, it calculates the energies of attraction and repulsion between related particles and predicts the general vitality of interaction. From this, deductions could be made as to the doubtless behaviour of the suspension. This is arguably an important query in pharmaceutical suspension formulation development, as a elementary specification for such a formulation is dose reproducibility, which is most easily achieved with a system which remains properly dispersed beneath all situations. It must also be remembered that every one particles will have some thermal vitality and will present some movement, whether attributable to Brownian movement, the results of gravity or exterior agitation. Particles within the main minimum zone present a better vitality of attraction than repulsion and are due to this fact more probably to transfer nearer together. Imagine two particles are simply far sufficient aside that the vitality of attraction balances out the power of repulsion, so the general power of interaction is zero. As they do so, the strength of the overall enticing forces increases, moving the particles nonetheless closer together, resulting in an additional enhance within the engaging forces, and so on. At first sight this would appear to be an excellent formulation strategy for pharmaceutical suspensions, as if the particles can be forced into the primary most zone, then they should remain impartial and hence dosing would be expected to be reproducible. At some point the particles will be sufficiently close such that the overall energy of interplay becomes adverse, i. In summary, due to this fact, formulating pharmaceutical suspensions in order that the particles are in the primary maximum zone can be considered to be risky. Depending on the kinetic energy of the particles, their behaviour will differ barely. As the particulate material inside a pharmaceutical suspension is the drug, the formulator has no alternative to change the physicochemical nature of the particles. Although theoretically the medium may be altered, which can then change the Hamaker fixed, most pharmaceutical suspensions, actually these supposed for oral drug supply, are aqueous. Hence the two elements in the suspension which contribute to the Hamaker constant (the drug and water) are fixed, and this factor is, in impact, not modifiable. The permittivity of the medium is said to its polarity, so subsequently various the medium will have a direct impact on the repulsive energy between particles in the system. Water is the most common medium for pharmaceutical suspensions, and addition of dissolved solids, corresponding to electrolytes, to water will have a comparatively minor effect on its permittivity, compared with the impact of changing the medium from water to , for example, oil. Overall, due to this fact, for the needs of pharmaceutical suspensions, the permittivity could be thought-about to be that of water and could have restricted variability. The distance between particles could be consid- Controlling particulate behaviour in suspensions From the previous dialogue, it can be seen that the behaviour of particles in suspension is advanced, even when solely two particular person interacting particles are thought-about, the behaviour in the end being dependent on the relative contribution of the repulsive and engaging energies at any separation distance. The interparticulate distance can be dependent on the focus of particles throughout the system, the next focus making it more probably that the particles might be physically located near each other. The Debye�H�ckel reciprocal length parameter is expounded to the gap over which the cost on the particle is decreased. It is dependent on the ionic energy of medium and can due to this fact be managed easily by the addition of ionizable supplies, such as sodium chloride. The radius of the particle (assuming sphericity) appears in both equations and so will have an result on each the attractive power and the repulsive power. It can be relatively easily controlled by the milling or micronization of larger particles to obtain a desired small particle dimension, or by crystal engineering strategies, intended to produce small particles directly from a solution. For instance, doubling the particle measurement will double all three calculated values, and halving the particle size will halve them. The effects could be rationalized by our considering the electrical double layer on particular person particles and how this impacts the interplay between particles. Addition of low to medium concentrations of ionizable materials will end in greater concentrations of the positively charged ion in the diffuse layer surrounding a particle (remember that the particle will carry a adverse charge in the absence of any absorbed materials on its surface), which can permit cost neutralization to occur over a shorter vary, i. This in turn signifies that the power barrier to escaping the secondary minimal is now higher and the particles will require a larger kinetic power to achieve this. As described previously, the secondary minimal is mostly considered to be desirable for pharmaceutical suspensions, as the particles will remain as free floccules rather than turning into aggregated, and so it follows that addition of low to medium concentrations of ionic materials will be helpful for pharmaceutical suspension formulations. However, the peak of the vitality barrier on the primary minimum will also be decreased, so if the particles have a excessive sufficient kinetic vitality. If the focus of the ionic material is sufficiently high, then a variety of the counterion costs will penetrate to the particle surface and cut back the general floor cost o and therefore the Stern potential, i. Hence, the particles stay drawn to one another at all size scales and are more likely to combination and coalesce. Adding surfactants to the suspension formulation at a stage under their cmc will outcome in their adsorption on the particle surface. However, the scale and magnitude of this impact might be depending on the chemical traits of the surfactant and addition of surfactant may end in an elevated or decreased chance of flocculation. When considering the consequences of movement, one should remember that deflocculated systems behave as particular person small particles, while flocculated methods behave as particular person large particles with a porous construction. Flocculated and deflocculated methods will present totally different particulate behaviour consequently. The overwhelming majority of pharmaceutical suspensions will contain particles in this dimension range, so sedimentation is a significant reason for particle motion. Here, the Stokes equation has been given in two equivalent varieties, defined by the particle radius and diameter: v= 2a2 g(- o) d 2 g(- o) = 9 18 (26. Because of their relative sizes, flocculated methods will settle rapidly, whereas deflocculated methods will settle more slowly.
Discount lanoxin 0.25 mg on-line
Other sugars or sugar alcohols hypertension kidney and dialysis specialists lanoxin 0.25 mg buy without a prescription, corresponding to glucose hypertension before pregnancy discount 0.25 mg lanoxin otc, sucrose, sorbitol and mannitol, have been used as different fillers to lactose, primarily in lozenges or chewable tablets, due to their pleasant style. Mannitol has a adverse heat of solution and imparts a cooling sensation when sucked or chewed. Apart from the sugars, maybe probably the most broadly used fillers are powdered celluloses of various sorts. Celluloses are biocompatible, are chemically inert and have good tablet-forming and disintegrating properties. They are appropriate with many medicine however, owing to their hygroscopicity, could additionally be incompatible with drugs susceptible to chemical degradation in the stable state. The most typical type of cellulose powder used in tablet formulation is microcrystalline cellulose. The name signifies that the particles have each crystalline and amorphous regions, relying on the relative position of the cellulose chains throughout the solid. The degree of crystallinity may differ relying on the supply of the cellulose and the preparation process. The degree of crystallinity will have an effect on the bodily and technical properties of the particles. Microcrystalline cellulose is prepared by hydrolysis of cellulose adopted by spray-drying. Depending on the preparation circumstances, aggregates of various particle dimension may be ready which have completely different flowabilities. A final essential example of a standard filler is an inorganic substance, dicalcium phosphate dihydrate. The substance can be obtained in both a nice particulate form, mainly used in granulation, and an aggregated kind. The latter possesses good flowability and is utilized in tablet manufacturing by direct compaction. Dicalcium phosphate dihydrate is slightly alkaline and will thus be incompatible with medication sensitive to alkaline conditions. Matrix former In order to affect or control the discharge of the drug from the pill, i. The matrix former is commonly a polymer or a lipid and may represent a significant fraction of the entire tablet weight. When the target is to increase drug dissolution, the matrix former could be a water-soluble substance or a lipid, and the drug is dissolved or suspended as fine particles in the matrix. When the target is to delay the drug launch, the matrix former could be both an insoluble substance (a polymer or a lipid) or a substance that forms a gel in contact with water. The drug is normally dispersed in particulate type within the matrix (for extra particulars, see later). Disintegrant A disintegrant is included within the formulation to be positive that the tablet, when in touch with a liquid, breaks up into small fragments, which promotes fast drug dissolution. Ideally, the pill should break up into particular person drug particles in order to produce the largest possible efficient surface area during dissolution. Deaggregation into drug particles will set up conditions for the quickest potential dissolution of the drug. The second step, granule disintegration or deaggregation, might end result in the formation of smaller aggregates or, if pushed to an ideal end point, discrete drug particles. The end level of the second step will in all probability depend upon a collection 528 of factors, such because the relative solubility and particle measurement of the drug and excipient particles of the granule. For instance, in a state of affairs where the drug particles are of low or very low solubility and are combined with a hydrophilic filler and binder of high solubility, the excipients could dissolve rapidly, leading to a dispersion of nice drug particles. During formation of a pill, the unique drug particles might fracture, and the resultant dimension distribution of the drug particles may thus not necessarily equate to that of the unique particles. Furthermore, the drug particles may also deform plastically throughout compression, which as nicely as might change their shape and look (see additionally later). However, as two primary processes are concerned within the disintegration event, the disintegrants to be used in plain tablets are categorized right here into two types: 1. These disintegrants act by facilitating the transport of liquids into the pores of the pill, with the consequence that the tablet could break up into fragments. One obvious sort of substance that may promote liquid penetration is surface-active agents. Such substances are used to make the drug particle surfaces more hydrophilic and thus promote the wetting of the stable and the penetration of the liquid into the pores of the tablet. It has also been instructed that different substances can promote liquid penetration, utilizing capillary forces to suck water into the pores of the pill. The further presence of a surface-active agent can also facilitate penetration of water into the pore system of the aggregates shaped throughout pill disintegration and thus enhance the deaggregation means of the aggregates too. Such a deaggregation and wetting course of may ideally produce a fantastic dispersion of discrete wetted drug particles that may enhance the surface area obtainable for drug dissolution and thus the rate of dissolution. Rupturing of tablets could be caused by swelling of the disintegrant particles during sorption of water. However, it has also been advised that nonswelling disintegrants can break the pill, and completely different mechanisms have been suggested. One such mechanism considerations a repulsion of particles involved with water and one other considerations the recovery of deformed particles to their authentic form in touch with water, i. Na stear fumar Mg stearate Ryoto 70 60 50 40 30 20 10 0 0 NaCl Teflon Na laur sulph. Lubricants are sometimes nice particulate substances, which are thus vulnerable to adhere to larger particles. In addition, research on the blending behaviour of magnesium stearate have indicated that this substance has the power to kind a movie which can cover a fraction of the surface space of the drug or filler particles (the substrate particles). A variety of elements have been suggested to affect the event of such a lubricant movie during mixing and therefore also have an result on friction and adjustments in pill properties, similar to the form and surface roughness of the substrate particles; the surface area of the lubricant particles; mixing time and intensity; and the sort and dimension of the mixer. Concerning the tablet-strength-reducing effect of a lubricant, other than the degree of floor coverage of the lubricant movie obtained during mixing, the compression behaviour of the substrate particles will also be essential. Drugs and fillers can thus be evaluated in terms of their lubricant sensitivity, i. An essential property for this lubricant sensitivity seems to be the degree of fragmentation the substrate particles undergo during compression (see later). To clarify the effect of lubricant film formation on the tensile energy of tablets, a coherent matrix model has been developed. This means that when a continuous matrix of lubricant-covered particle surfaces exists in a tablet, alongside which a fracture plane can be fashioned, the strength of the pill is significantly decrease than that of tablets formed from unlubricated powder. Many powders are vulnerable to adhere to the punches, a phenomenon (known in the pharmaceutical industry as sticking or picking) which is affected by the moisture content of the powder.

